 
Thank you so much for the explanation, but there was a mistake there as mdfenko states, right? Because I also think if you dissolve 10g in 1000ml that's 1%, not 10%. Hope that helps without too much headache. 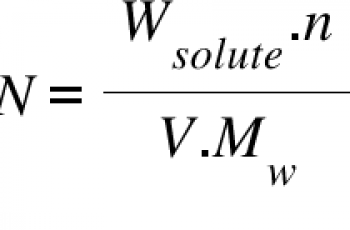
Especially so in toxicology, environmental science and chromatography). (You also sometimes see ppm, part per million or parts per thousand. Use w/v when the solutes are solid and vol/vol when they are liquid. So a 5% glycerol solution contains 5ml glycerol in 95ml water (total volume is 100ml). (In fact, you would suspend the 10g in solvent and make up to 1l.which might not be exactly 1000ml depending on the solute's density).Ī 5%w/w solution of glucose would have 5g glucose and 95g water (in fact, 95ml since the density of water is 1g/ml). Plug in your values and solve the equation to find the concentration of your solution. Set up your equation so the concentration C mass of the solute/total mass of the solution. %w/w.įor a 10%wt/vol solution you take 10g of solute mass and suspend it in 1000ml. Divide the mass of the solute by the total mass of the solution. Percent concentration can be expressed in other ways too. SmoochiePie, wt/vol is just a way of expressing weight by volume concentration. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
